Key Steps in Plastic-to-Oil Pyrolysis

The global plastic waste crisis has reached critical levels, with over 400 million tons of plastic produced annually and less than 10% effectively recycled. Pyrolysis—the thermal decomposition of plastic in the absence of oxygen—has emerged as a promising technology to convert waste plastic into usable fuel oil. Here's how the process works and what investors need to know.
Collection & Cleaning
The pyrolysis process begins with sourcing clean, dry plastic waste. Not all plastics are suitable for conversion:
- Optimal plastics: Polyethylene (PE) and polypropylene (PP) deliver the highest oil yields
- Acceptable: Polystyrene (PS) can be processed but produces different output ratios
- Avoid: PET bottles, PVC, and mixed contaminated plastics produce toxic byproducts or low-quality oil
- Facilities must implement rigorous sorting systems to remove contaminants including glass, metal, paper, and food residue. Even small amounts of moisture can significantly reduce efficiency and increase energy costs.
- The Heating Process
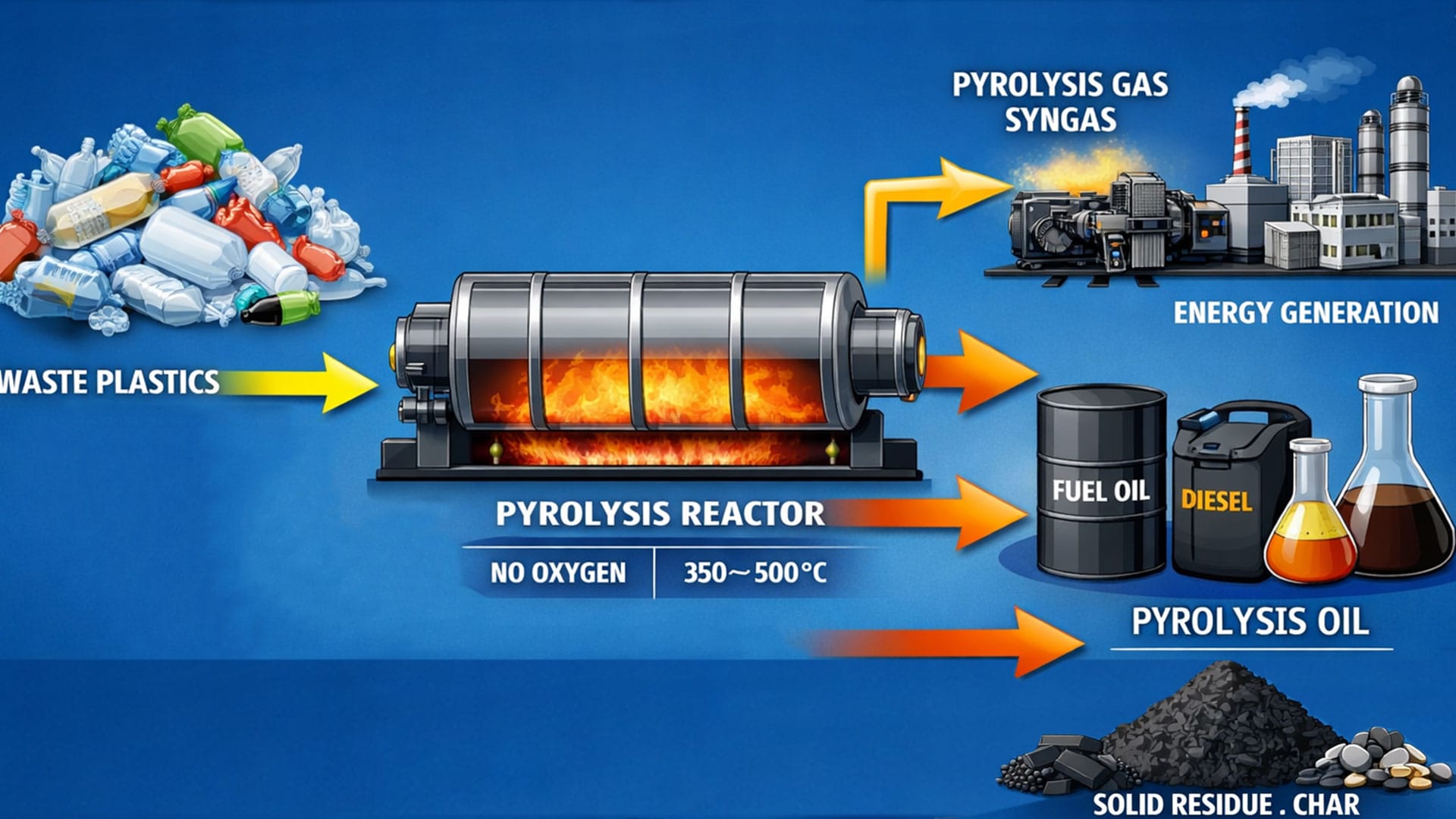
- Sorted plastic enters a sealed reactor or chamber where it's heated to temperatures between 400–800°F (200–430°C). Without oxygen present, the plastic cannot burn. Instead, the polymer chains break apart through thermal decomposition.
- Temperature control is critical for output optimization:
- 400–500°F (200–260°C): Maximizes oil production, ideal for PE and PP
- Above 500°F (260°C+): Shifts output toward gaseous hydrocarbons (methane, propane)
- Industrial facilities often employ catalysts such as zeolite or silica-alumina to lower required temperatures and improve oil yield by 15–25%. This reduces energy costs while increasing throughput.
- Output Products
- A typical pyrolysis operation produces three distinct outputs:
- Pyrolysis Oil (60–80% of output):
- A crude oil-like substance with a calorific value of approximately 40 MJ/kg, comparable to commercial diesel. This oil can be sold directly to industrial burners or refined further into gasoline, diesel, or kerosene.
- Synthetic Gas (10–20%):
- A mixture of methane, ethane, propane, and other hydrocarbons. Most facilities recycle this gas to fuel the pyrolysis reactor, creating a partially self-sustaining energy loop.
- Carbon Char (10–20%):
- Solid residue that can be sold as fuel briquettes, activated carbon for filtration, or as a filler material for construction products.
- Refining Requirements
- Pyrolysis oil is not ready-to-use fuel. The crude output contains impurities including chlorine, sulfur, and oxygen compounds that must be removed before the oil meets commercial fuel standards.
- Refining steps include:
- Distillation: Separates hydrocarbons by boiling point into gasoline, diesel, and heavy fuel fractions
- Hydrotreatment: Removes sulfur and nitrogen compounds using hydrogen
- Filtration: Eliminates particulate matter and remaining contaminants
- For most operations, selling crude pyrolysis oil to refineries is more economical than building in-house refining capacity.
- Economic & Environmental Considerations
- Conversion Efficiency:
- Industrial-scale pyrolysis facilities achieve 70–90% oil conversion rates. Small-scale operations typically see 50–65% efficiency due to heat loss and less precise temperature control.
- Energy Requirements:
- The process is energy-intensive, consuming approximately 8–12 kWh per kilogram of plastic processed. Facilities that capture and reuse synthetic gas can offset 40–60% of energy costs.
- Capital Costs:
- A commercial pyrolysis plant processing 10 tons per day requires $2–5 million in initial investment, depending on automation level and emission control systems.
- Environmental Trade-offs:
- While pyrolysis diverts plastic from landfills and oceans, it is not carbon-neutral. The process produces CO₂ emissions from both energy consumption and the breakdown of petroleum-based plastics. Life cycle assessments show pyrolysis produces 30–50% fewer emissions than incineration but 2–3x more emissions than mechanical recycling.
- Market Outlook
- The global plastic pyrolysis market is projected to reach $15.8 billion by 2030, growing at 14.2% annually. Key drivers include:
- Stricter landfill regulations in the EU and North America
- Corporate commitments to circular economy principles
- Rising crude oil prices improving pyrolysis economics
- Extended Producer Responsibility (EPR) laws forcing brands to fund recycling infrastructure
- Major oil companies including Shell, BP, and TotalEnergies have announced pyrolysis partnerships, signaling mainstream acceptance of chemical recycling as part of the energy transition.
- Investment Implications
- Pyrolysis represents a convergence of waste management, energy production, and environmental technology. For investors, the sector offers exposure to:
- Waste management companies expanding into chemical recycling
- Specialized pyrolysis technology providers selling equipment and catalysts
- Refineries integrating pyrolysis oil into existing processing infrastructure
- Private project developers building regional processing facilities
- Key risks include regulatory uncertainty around emissions standards, competition from mechanical recycling improvements, and potential carbon taxes on pyrolysis outputs.
- Sources:
- International Energy Agency, "Chemical Recycling Market Assessment" (2025)
- McKinsey & Company, "The Future of Plastic Waste Processing" (2026)
- Global Pyrolysis Oil Association, "Industry Production Standards" (2025)
- Environmental Science & Technology Journal, "Life Cycle Analysis of Plastic Pyrolysis" (2025)




